The reason this distinction is so important to make is that organic chemistry deals with such a broad range of compounds, despite the fact that carbon is but a single atom. Such is the case with the division between organic and inorganic substances, for which there is no comprehensive rule. Chemistry, much like music and astronomy, is riddled with peculiar historical nomenclature. 
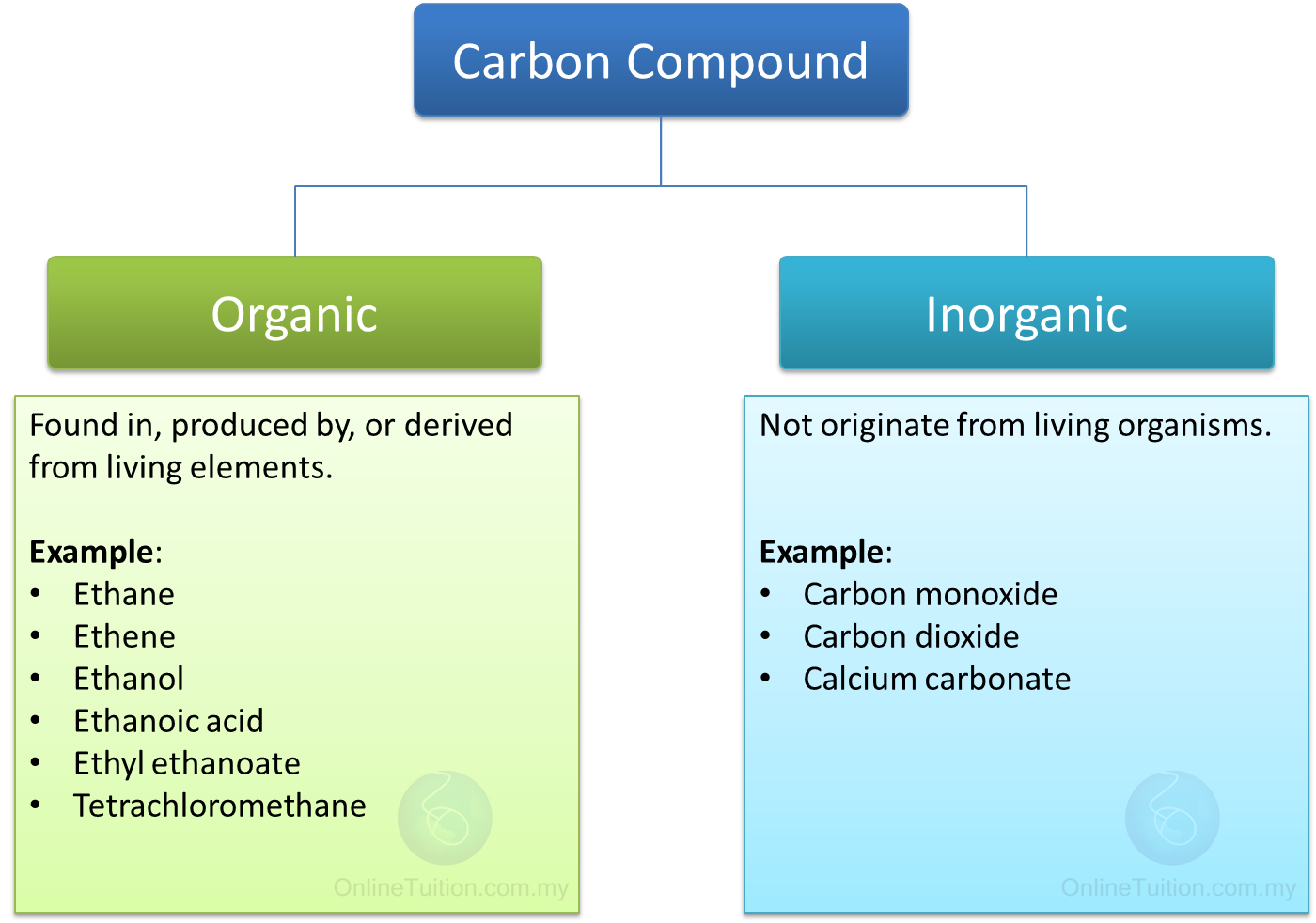
Though an inorganic compound may contain either hydrogen or carbon, containing both generally makes it organic.  
Inorganic chemistry deals with all the other parts of the periodic table. The easiest way to describe the difference between inorganic chemicals and organic chemicals is that organic chemistry covers compounds that are based on carbon and hydrogen combinations, and may or may not contain oxygen. Examples of inorganic compounds and substances.The differences between organic and inorganic substances.In this article, we will cover how inorganic compounds compare to organic compounds, some examples of important inorganic substances, and different applications and industries where they are produced and used. These are known as organometallic compounds. While materials like minerals and metals fit tidily into this definition, there are also plenty of inorganic compounds in which a metalloid or metal is bonded with carbon. The term "inorganic" refers broadly to compounds that do not contain both carbon and hydrogen. They form useful acids, bases and inert materials utilized for their specific attributes such as conductivity, catalysis, and reactive chemistry. Created in naturally, synthetically, and industrially, inorganic chemicals are responsible for the fields of semiconductors, pigmentation, coating and surfactants, fuel, medicine, and chemical manufacturing. Inorganic chemicals are most abundant in nature, and are the basis for many vital industries.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
